This may not sound terribly interesting at first, but it proves problematic for proteins that are in awkward places, for example in the outer membrane of Gram negative bacteria. These outer membrane proteins (OMPs) not only have to fold up correctly inside the membrane but they have to actually get to the outer membrane In Gram negative bacteria, this means first getting through the inner membrane, then across the peptidoglycan layer between the membranes, and finally half way through the outer membrane in order to coil up correctly inside it.
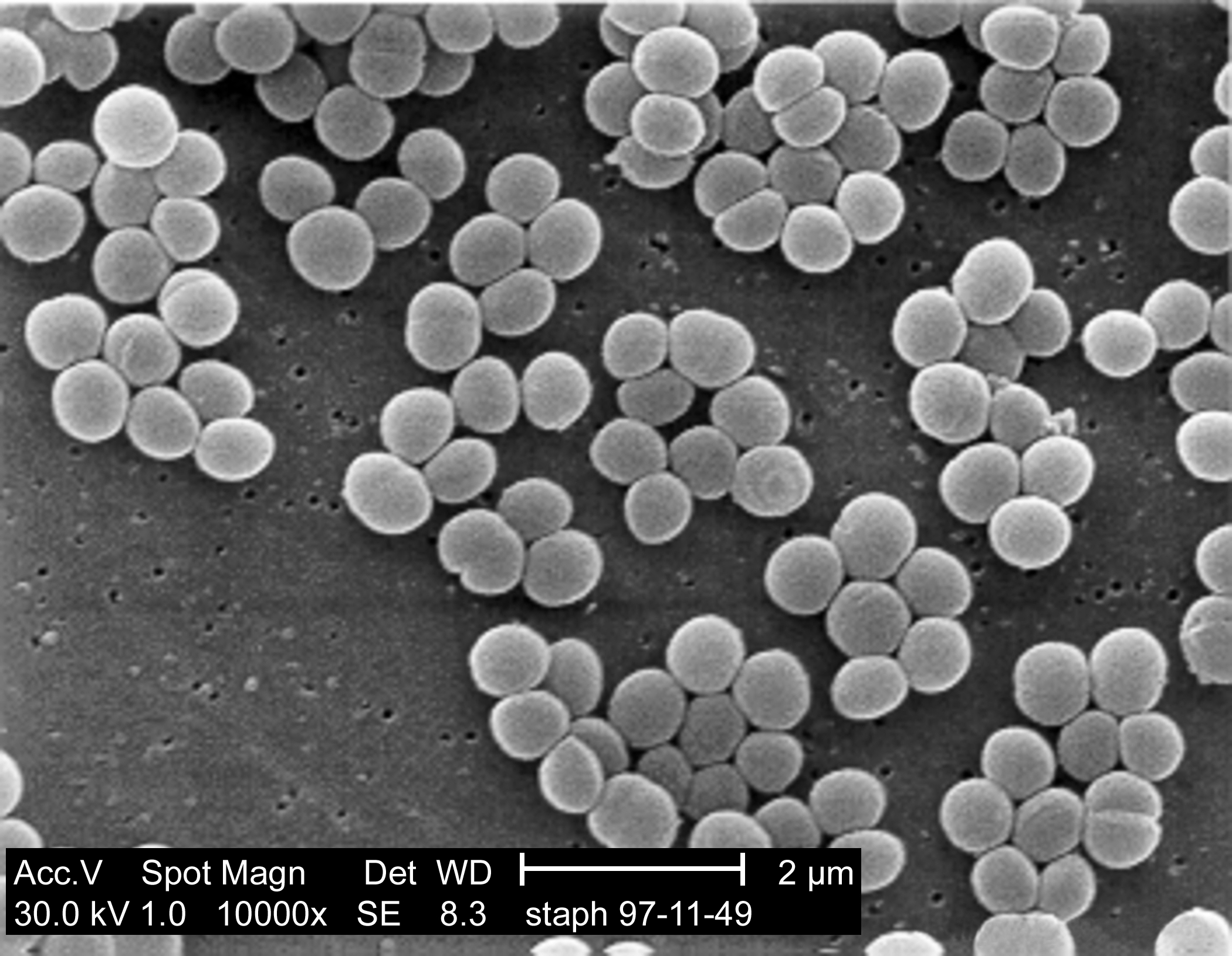
The type of proteins found inside the outer membrane are usually B-barrel proteins, so called because they contain lots of protein folds known as a B-sheets
 , which can wrap up to form a channel shape as shown in the example on the right. Each blue arrow is a single B-sheet and these fold specifically to form pore-like structures which in the case of porins make a little hole through the membrane.
, which can wrap up to form a channel shape as shown in the example on the right. Each blue arrow is a single B-sheet and these fold specifically to form pore-like structures which in the case of porins make a little hole through the membrane.Transport of B-barrel OMPs accross the inner membrane is achieved by synthesising them with a signal sequence attached to one end. This signal sequence is recognised by proteins on the inner membrane and ATP energy is used to pump the proteins accross the inner membrane and into the periplasm (the space between the two membranes). Once in the periplasm they bind to little chaperone proteins which carry them safely to the complex responsible for folding them correctly into the outer membrane, the rather awesomely named BAM complex.
 As an aside the chaperones do have to get the OMPs there fairly promptly as there are proteases that float around in the periplasmic space and degrade any proteins that don't get incorporated into the outer membrane quick enough.
As an aside the chaperones do have to get the OMPs there fairly promptly as there are proteases that float around in the periplasmic space and degrade any proteins that don't get incorporated into the outer membrane quick enough.One of the key proteins in the BAM complex is BamA as knocking it out results in a lot of unfolded OMPs in the periplasm (and probably a field day for the proteases). BamA consists of two major components, a B-barrel domain which anchors it into the outer membrane, and five "polypeptide transport-associated" domains, shortened to POTRA by someone who didn't like three-lettered acronyms. The POTRA domains do what they say, they are associated with the transport of proteins (polypeptides).
It's still a little uncertain quite how the BAM complex works but a couple of the presentations on the topic were convering it, including work done on changing the genes between different bacterial species. All Gram-negative bacteria have a BamA gene, however taking the BamA gene from one bacteria and putting it into another does not end happily unless it's done between two very close species. Closer research with chimeric proteins (i.e proteins that are half from one bacteria and half from another) shows that this only applies to the POTRA domains. The anchoring B-barrel can be switched between several different species, but the POTRA domain is very species specific.
Another interesting thing to address is how BamA gets itself into the outer membrane. One of the periplasmic chaperone proteins, Skp, is thought to be involved in this process, and it was found that when the outer membrane was negatively charged Skp is involved in inserting BamA into the membrane, whereas when the negative charge is removed Skp inhibits BamA folding and insertion. Negative charge is caused by an increase in the phosphatidylglycerol content in the membrane. I found that idea quite exciting as it implies that the bacteria can control where they want the BAM complex to go. The idea of membranes forming "lipid rafts" with certain components that organise where proteins are held is not a new one, and the BAM complex forming in specific places in order to create the correct outer membrane protein concentration is one that appeals to me.
They may just be single little cells with no true nucleus, but they are capible of a lot of control over their intracellular processes!
---
Knowles TJ, Scott-Tucker A, Overduin M, & Henderson IR (2009). Membrane protein architects: the role of the BAM complex in outer membrane protein assembly. Nature reviews. Microbiology, 7 (3), 206-14 PMID: 19182809
Johnson, A., & Jensen, R. (2004). Barreling through the membrane Nature Structural & Molecular Biology, 11 (2), 113-114 DOI: 10.1038/nsmb0204-113
---
Follow me on Twitter!